New approach methodologies in human regulatory toxicology – Not if, but how and when! - ScienceDirect
Por um escritor misterioso
Descrição

Regulatory assessment of chemical mixtures: Requirements, current approaches and future perspectives - ScienceDirect

New approach methodologies in human regulatory toxicology – Not if, but how and when! - ScienceDirect

Acceptable Intakes (AIs) for 11 Small molecule N-nitrosamines (NAs) -Pub - New Scientific Knowledge & Development - Nitrosamines Exchange

Paving the way for application of next generation risk assessment to safety decision-making for cosmetic ingredients - ScienceDirect
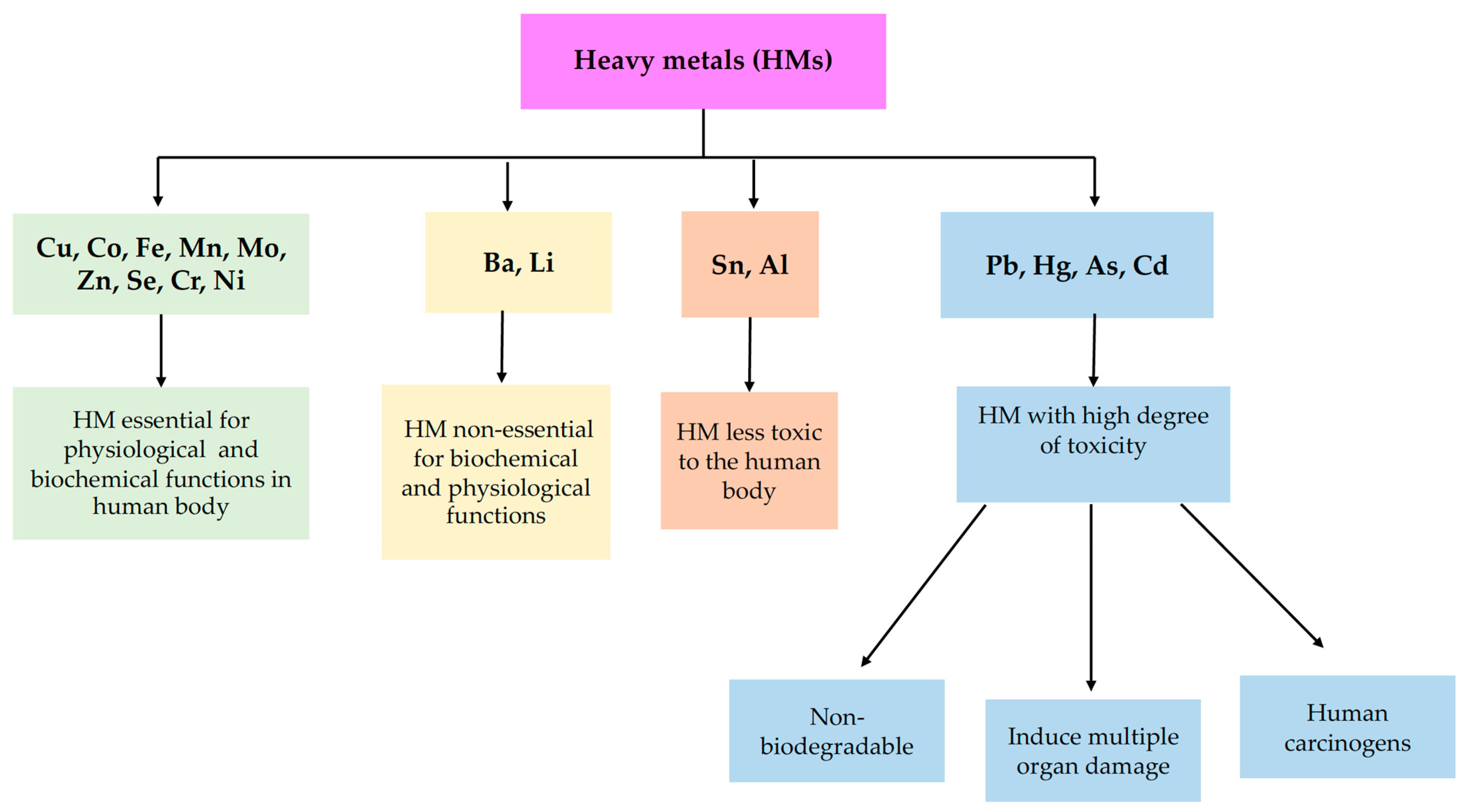
Toxics, Free Full-Text

New Approach Methodologies (NAMs) for safety testing of complex food matrices: A review of status, considerations, and regulatory adoption - ScienceDirect

Comparison of regulatory pathways for the approval of advanced therapies in the European Union and the United States - Cytotherapy

An evaluation framework for new approach methodologies (NAMs) for human health safety assessment - ScienceDirect

Regulatory Toxicology and Pharmacology, Vol 137, January 2023

Assessing Toxicity with Human Cell-Based In Vitro Methods: Trends in Molecular Medicine

Principles underpinning the use of new methodologies in the risk assessment of cosmetic ingredients - ScienceDirect
New approach methodologies (NAMs) for human-relevant biokinetics predictions

Toxicogenomics: A 2020 Vision: Trends in Pharmacological Sciences

Suspect and non-targeted screening of chemicals of emerging concern for human biomonitoring, environmental health studies and support to risk assessment: From promises to challenges and harmonisation issues - ScienceDirect

Testing the feasibility of a new way of toxicity testing and reduction
de
por adulto (o preço varia de acordo com o tamanho do grupo)







